Image Quality Explained

A Snapshot Serengeti Camera-trap image
Those of you who have been with us for some time will probably have noticed that the image quality since we switched to the Snapshot Safari platform has reduced, sometimes dramatically. Before I go any further, we are trying hard to fix this but in the meantime I thought I would try and explain what the issues are in a hope that it may induce a little more patience from you. I am afraid that I really am technically challenged when it comes to computer stuff so I am going to be a little vague here but please, if there is anyone out there with more knowledge who can either help explain more appropriately or better still offer our team help don’t hesitate to get in touch.
So the trouble all started when Snapshot Serengeti joined the bigger Snapshot Safari platform at the start of this year. At this time Zooniverse was having a big overhaul with older projects operating on Ouroboros moving over to the Panoptes format. Essentially Ouroboros and Panoptes are both software packages which enable projects to build their pages and run them.
Of course Snapshot Serengeti being one of the oldest Zooniverse projects was designed using Ouroboros and has had some teething problems with the switch over. One thing to remember is that the teams involved with bringing all the camera trap images to the Snapshot Serengeti platform are for the most part unpaid graduate and undergraduate students studying ecology. They are not experts in computer programming yet have to keep the platforms running and fix all the problems.
In the old days the University of Minnesota based team would upload the batches of images from the camera traps and send them to Zooniverse who would process and upload them to the platforms. That was when there were a dozen or so projects. There are now over 50 active projects. Can you imagine how long it would take for Zooniverse to do all the uploading? To address this problem they have asked individual projects to manage the uploading themselves. To complicate this process a little more they have also placed a 600GB maximum file size on the images.
This all means that the team of ecologists at Minnesota have to engage computer code developers to write custom scripts enabling their super computers to interact with the Zooniverse web platform. The image quality issue then is not because we have started using different camera’s or taking images at a lower resolution it is due to the code that compresses the images from their full size to less than 600GB. Those images that were smaller in the first place have been less effected than the larger ones and hence the mixture of quality that we are seeing.
So as I said earlier we are trying hard to get this problem sorted and bring you back the kind of top rate images you are used to and hope to have things sorted with the next batch of images we upload. In the meantime please spare a thought for the team and remember that like you they are all volunteers, all be t with a slightly more vested interest in the research project. I hope that you will bear with us and keep up the much needed support you have always given us.
The Giraffe and the Oxpecker

Those of you who follow our Facebook page will have seen recently that Meredith Palmer, one of Snapshot Serengeti’s scientists and PhD candidate with Minnesota University just published a paper in African Journal of Ecology with the catchy title;
Giraffe Bed and Breakfast: Camera traps reveal Tanzanian yellow-billed oxpeckers roosting on their large mammalian hosts.
The paper highlights one of the more unusual behaviour traits documented by our cameras and discovered by our classifiers of yellow-billed oxpeckers (Buphagus africanus) roosting on giraffe at night time.
Those of you that have been with us a while may have had the pleasure of finding one of these night time images of giraffe with oxpeckers tucked up safe and snug between their back legs. In fact I wrote a blog about this back in 2014.
https://blog.snapshotserengeti.org/2014/01/17/the-curious-case-of-the-giraffe-and-the-oxpecker/
Two species of oxpecker are found in the Serengeti, the red-billed and the yellow-billed oxpeckers. Whilst the red-billed will feed from a wide range of hosts from impala and wart hog to hippos the yellow-billed oxpecker is more discerning and prefers large hosts such as buffalo, eland and giraffe. The problem with this choice is that these animals are far roaming and if the birds were to find trees to roost in at night, and these can be sparse in the Serengeti, the yellow-billed oxpecker could struggle to locate its host the following morning. It seems they have overcome the problem by staying over on the hosts. What’s more is these clever birds have opted for the premium rate rooms where they are not disturbed during the night for, as is well documented, giraffe almost never lay or sit down at night time preferring to stay upright.
So although during the day yellow-billed oxpeckers are found on several large mammal hosts most of the night time images are of giraffe roosts. It seems they also have a preference for the groin area of the giraffe. It is not hard to imagine that this would be the warmest safest spot on the giraffe, the cavity created where the two hind legs meet is spacious enough to accommodate a small flock of birds and of course is also very attractive to ticks so if they fancied a mid-night snack…..
It is these unexpected discoveries that make the project so exciting and worth all our effort in taking part so next time you are racing through the classifications take a little time to have a closer look at the images, you never know what is waiting to be discovered.
If you want to read more about Meredith’s paper you can read the following:
https://news.nationalgeographic.com/2018/02/animals-serengeti-tanzania-birds/
Welcome New And Old

Lion pride
So the new Snapshot Safari base camp for Snapshot Serengeti is a month old and teething problems aside all seems to be going well. I just wanted to take this opportunity to welcome all our new classifiers and to say a big thanks to all our old classifiers who have stuck with us. But most of all a massive thank you to our moderators who have worked so hard to make the transition run so smoothly. They have answered all your questions and queries without my back up due to the unfortunate timing of my own African field trip falling during the launch of Snapshot Safari.
It is not the first time Snapshot Serengeti has seen a big change. Some of you may remember its first outing as Serengetilive back in 2011. In those days things where a lot slower, you started classifying by first choosing an individual camera and working through it. There was an option to skip images, leaving them for someone else. Of course what ended up happening was all the hard to identify images and all the no animal grassy images were left to the end so that some people never got the chance to classify any animals.
We then progressed, in 2012, onto the Zooniverse platform and saw a huge change to the way things worked. Suddenly there was a lot more interaction between the scientists and the community. This was when the famous algorithms where developed by Margaret Kosmala and Ali Swanson and their team to act as a fail proof to anyone incorrectly identifying images.
We are all very grateful for their hard work and dedication that results in us classifiers being confident that our guesses won’t mess everything up.
So I hope that you are enjoying this third incarnation of Snapshot Serengeti and can be proud that it has worked so well over the years that it has spawned so many new projects.
My own field trip to Africa is coming to an end this week and I will be back in the land of internet connection. I will then hopefully be bringing you more regular posts and more updates on the project itself and how it is progressing. In the meantime don’t forget to check out our facebook and twitter pages.
Dynamic Landscape of Fear

The Snapshot team have written another paper using the Snapshot data we all help to classify. The paper A ‘dynamic’ landscape of fear: prey responses to spatiotemporal variations in predation risk across the lunar cycle can be found at http://onlinelibrary.wiley.com/doi/10.1111/ele.12832/full for those of you interested in reading the original.
Lead by Meredith Palmer the paper explores how four ungulate species, buffalo, gazelle, zebra and wildebeest respond to predation risk during differing stages of the lunar cycle. These four make up the bulk of the African lion’s diet in the Serengeti along with warthog. Of course warthog are strictly diurnal so are not affected by the lunar cycle as they are tucked up nice and snug in a burrow.
For the other four night time can be a stressful time. None of these animals sleep all night, they snatch rest here and there, keep grazing and most importantly of all keep a watchful eye or ear out for possible attack.
It has long been thought that prey species territory is shaped by fear and that animals have safe areas (where they rest, give birth, etc) and risky areas where they instinctively know predators may be lurking. These areas trigger a risk versus reward response as they often contain better forage/water etc.
What Meredith and the team argue is that this landscape of fear is very much dynamic changing not only with seasons and night and day but on a very much finer scale as influenced by light availability through the moon.
Lions find it so much easier to hunt during nights where the moon gives of least light. It gives them a great advantage to stalking close to their prey using the dark as a kind of camouflage. The prey species, on the other hand, are at a distinct disadvantage, they can’t see the stalker and even if they sense its presence they are reluctant to flee as this presents a great risk in itself if they can’t see.
Meredith and her colleagues took the data from Snapshot Serengeti to quantify nocturnal behaviour of the key species using the presence or absence of relaxed behaviour (defined when we classify a species as resting or eating.) They then overlapped this with data collected through Serengeti Lion Project on lion density and hunting success. This data enabled them to work out what areas where high or low risk to the prey species. Using a clever statistical program, R, the data was analysed to see if lunar cycle had any bearing on animal behaviour, in particular, predator avoidance.
They found that moonlight significantly affected the behaviour of all four species but in a variety of ways. As we mentioned before there is often a good reason to venture into the high risk areas and the trade off in increased risk of predation is a really good feed. Buffalo for instance don’t change their use of space so much but were found to form more herds on dark nights. It seems safety in numbers works well for buffalo. Zebra react similarly in their herding activity but are much more erratic when it comes to space use, moving around a lot more randomly keeping potential predators on their toes.
Each species showed an aversion to using high risk areas at night but, particularly wildebeest and zebra, were found to increase their use of these areas when the moons luminosity was higher and safety increased. It was noted that high risk areas where avoided more frequently in the wet season than the dry. The thought being that there is increased hours of moonlight during the dry season that the animals take advantage of. Perhaps too the drive to find enough good food is a factor.
This paper serves to remind us that although what we do at Snapshot Serengeti is fun it is more than just a way for us classifiers to pass the time. It really has a very significant role in science and that role is ever increasing.
Progress and stuff
Some of you will have noticed that our progress bar on season 10 has not been showing any progress. Well it turns out that we have made loads of progress, it’s just the bar that was not getting anywhere.
The good folks at Zooniverse have fixed it for us and you will now see we are about half way through season 10 which is fantastic. There are just under 700 000 images to classify this season so thanks to you, are dedicated team of citizen scientists we have around 350 000 left to go. That’s 350 000 chances of finding that one image you have been waiting for. I have noticed recently lots of you posting on talk that you have classified your first ‘waterbuck’ or ‘serval’. If you haven’t discovered your dream find yet there is still time and yes there is a season 11 in the wings.
Whilst on the subject of talk I wanted to gently remind everyone of a few etiquette points.
#Hashtags, love ‘em or hate ‘em they are part of social media and they are not going away. On Snapshot Serengeti we use them for a specific reason and that is to help others to search for and find certain images.
If you have found a great image that you think others will want to see and you are certain of the species then go ahead and hashtag it, but, if you find an image that you are not sure of then please don’t hashtag it with your guess. You can still put the pictures up in talk for discussion and perhaps someone else will be along who is positive about the id and can then hashtag it. Basically, please use hashtags thoughtfully.
Which brings me to another point; if you can’t identify an image and you post it up for discussion always give us your best guess. No one will laugh; it’s what makes it fun seeing what other people make of the images when you are really stumped. Many a time I have confidently shared a tricky image almost certain for instance it’s a long sort after rhino only to have someone else’s eyes point out that if I look a bit closer that actually it is a rock! Even our expert modifiers get things wrong occasionally and are reluctant to confidently make a call on certain images. Some of them are just so darn impossible to id. So just give it your best shot, it’s what everyone else does.
The main aim is to enjoy yourself, challenge yourself and use other peoples experience when yours fails you. The Snapshot family of classifiers and moderators is a dedicated and knowledgeable bunch and as I have said before, this project would not exist without you all. Keep up the great work one and all.
Citizen Science Conference

Meredith giving a project slam Photo: Avi Baruch
Meredith has been busy this past week attending the Citizen Science conference in St Paul, Minnesota. She reports back that it was a fantastically stimulating conference that confirms the high esteem that citizen science has grown within the science community.
The yearly conference sees a diverse group of people from researchers, educators and universities to the likes of NGO’s and museums get together to discuss the use and promotion of citizen science. Although we at Snapshot Serengeti have been aware of its great impact for some time citizen science is now emerging and is recognised as a powerful tool in the advancement of research by many.
Those attending the four day event collaborated by sharing their varied experience and ideas on a variety of topics. The collection and sharing of data and how to impact policy was discussed. There was focus on how to use citizen science as an engaging teaching tool, how to bring citizen science to a wider audience and how to involve citizens more in research. Those attending brought their joint experience and expertise together to discuss how citizen science impact on science could be measured and evaluated. If you want to find out more about the conference then visit this link.
http://citizenscience.org/association/conferences/citsci2017/
We sometimes forget when working away at classifying our stunning images on Snapshot Serengeti that there is a lot of tech going on that enables us citizen scientists to be of use to the scientists. Meredith gave what’s known as a ‘project slam’ essentially a 5 minute presentation about our work on Snapshot Serengeti and how it has paved the way for helping other cameratrap citizen science projects. A quick look around Zooniverse will show just how many there are now.
The massive amount of data produced over several seasons through Snapshot Serengeti have allowed the development of a robust, tried and tested methodology that smaller projects would have taken years longer to develop. Just contemplate the work that went into developing interfaces, protocols, pipelines and algorithms for taking millions of classifications of untrained volunteers and turning them into a dataset which has been verified to be >97% accurate.
It is awesome to see that something we all find so truly engaging can translate into such serious stuff in the field of science. I think we, the citizen scientists, and the Snapshot team can be rightly proud of our work on this brand new branch of science
Topi versus Hartebeest
Here is another pair of antelope that are often muddled up on Snapshot Serengeti; topi and hartebeest. These two share a similar size and body shape and for those of you not familiar with them they can prove a bit tricky.
Topi and hartebeest belong to the same tribe, Alcelaphini, which also includes wildebeest. These antelope typically have an elongated face, long legs, short necks and stocky bodies. Although these antelope have reasonably large bodies their long legs mean they have retained the ability to run fast, a good adaptation for life on the open plains. It is believed that the long face developed in place of a long neck in order to reach the grasses they consume.
There are several species of both topi and hartebeest in Africa, two are found in the Serengeti. Coke’s hartebeest or kongoni (Alcelaphus cokii) are selective grazers with browse making up less than 4% of their diet. Serengeti topi (Damaliscus jimela) are 100% grazers
In both species males are territorial but topi also form leks from which to display to passing females. Males holding territory close to the lek are more desirable to females. Dominant females will actively prevent subordinate females from mating with these males.
Topi Hartebeest
So side by side we can see that the topi is much darker coloured than the hartebeest with distinct sandy socks up to its knees and conspicuous black patches on the thighs and shoulders. In contrast the hartebeest has pale legs and underbelly with a darker upper body. The paleness forms a patch on the top of the thigh.
Topi Hartebeest
From behind the contrast between leg colour and backside is very obvious with topi sporting dark legs with pale rump and back and hartebeest pale legs and rump with dark back.
Horn shape is also different. A topi’s horns sweep up and back whereas a heartebeest’s sweep out to the side before kinking back. They also sit on a prominent bony ridge on the top of the head.
Hopefully this will help you tackle all the images waiting on season 10.
The Data Game

Team member checking a camera-trap
So we all know there are millions of images on snapshot Serengeti and that it is us citizen scientists who do all the work classifying them. The scientists can then get on with the task of figuring out what’s going on out there in the animal kingdom, hopefully in time to save some of it from our own destructive nature.
But… have you spared much thought as to how the images go from over 200 individual camera-traps dotted around the Serengeti to the Zooniverse portal in a state for us to start our work.
Firstly the SD cards have to be collected from the cameras and as this is an ongoing study replaced with fresh SD cards. This is done about every 6 to 8 weeks. A camera traps batteries can actually go on performing far longer than this but as the field conditions can be tough you never know when a camera may malfunction. This time frame is a good balance between not ending up with months worth of gaps in the data and not spending every minute in the field changing cards.
The team are able to check about 6 to 10 sites a day so with 225 cameras in play it takes around a month just to get to each site. Mostly the cameras are snapping away happily but there are always some that have had encounters with elephants or hyena but actually some of the most destructive critters can be bugs, they like to make nests of the camera boxes. As well as checking the cameras themselves the sites need to be cleared of any interfering foliage, we all know how frustrating a stray grass blade can be.

Snake camping out inside the camera-trap
So with a hard drive full of all the data it then has to wait for a visiting field researcher to hand carry it back to the University of Minnesota, USA. It means the data is only received every 6 months or so but it is far safer than trusting the post. Once safely received it is up to Meredith to start the painstaking work of extracting the date time stamps. As sometimes happens there are glitches and she has to fix this by figuring out when the camera went off line or when capture events got stuck together. She says it is much like detective work. The images are then assigned codes and stored on the Minnesota Supercomputer Institute (MSI) servers.
Once it is all cleaned up and backed up it is sent to the Zooniverse team who then format it for their system giving new identifiers to each image. Finally it is ready for release to all the thousands of classifiers out there to get to work on.
So as you can see it really is a team effort and a massive under taking. It is no good collecting tonnes of data if there is no one with the time to do anything with it. I will take this opportunity again to thank you for all your help with the project. Keep up the good work.
Why we do it
Congratulations, your time classifying images on Snapshot Serengeti has resulted in yet another scientific paper. Over 70,000 of you have contributed to analysing the millions of images produced by the 225 Snapshot Serengeti cameras over the last few years. Thanks to all your effort the cameras are still rolling, creating one of the longest running cameratrap studies going. This data set is so important to scientists because of the size of the area it covers as well as the length of time it has been recording for. It allows them to ask many and varied questions about a naturally functioning healthy ecosystem and in today’s changing world it has never been so important to figure out what makes this planet tick.
The paper ‘The spatial distribution of African Savannah herbivores: species associations and habitat occupancy in a landscape context’ was published last year in Philosophical Transactions B. Visit here to read the article.
http://rstb.royalsocietypublishing.org/content/371/1703/20150314
The Snapshot Serengeti team argue that if we want to predict the impact of changes/ losses of large mammals in the future we need to have a quantitative understanding of a currently functioning ecosystem. It just so happens that the Snapshot data set is perfect for this. The Serengeti National Park is representative of the grass dominated Savannahs of East Africa which are home to the world’s greatest diversity of ungulate (hoofed animals) grazers.
The team present a neat graphic that shows how the various elements interact to affect herbivore habitat occupancy.

Predators, herbivores, termites, fire, grasses and trees all play a role in determining where different herbivores choose to roam.
It seems that herbivore body size is also important to habitat selection. For example large herbivores survive by bulk grazing whereas small herbivores concentrate on grazing quality over quantity. Recently burned ground results in new vegetation growth. This growth is relatively high in nutrients compared with unburned patches and the same can be found on and around termite mounds. Small herbivores were found to occupy these areas but the sparse coverage does not favour large herbivores that must eat more volume.
The paper highlights the complex relationship between predators, herbivores, vegetation and disturbance and is well worth a read. Next time you are classifying images see if you agree. Do you see many herds of zebra or wildebeest on burnt areas or is it mostly Thompson’s gazelle? It’s another way to look at the images you classify.
Getting Good Data, Part II (of many)
Okay, so by now you’ve heard dozens and dozens of times that you guys produce really good data: your aggregated answers are 97% correct overall (see here and here and here). But we also know that not all images are equally easy. More specifically, not all species are equally easy. It’s a lot easier to identify a giraffe or zebra than it is to decide between an aardwolf and striped hyena.
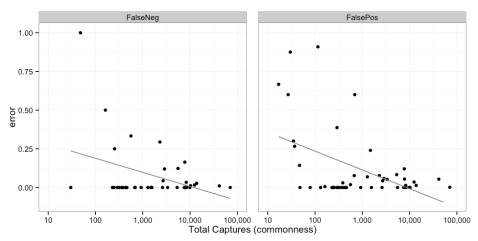
The plot below shows the different error rates for each species. Note that error comes in two forms. You can have a “false negative” which means you miss a species given that it’s truly there. And then you can have a “false positive,” in which you report a species as being there when it really isn’t. Error is a proportion from 0 to 1.
We calculated this by comparing the consensus data to the gold standard dataset that Margaret collated last year. Note that at the bottom of the chart there are a handful of species that don’t have any values for false negatives. That’s because, for statistical reasons, we could only calculate false negative error rates from completely randomly sampled images, and those species are so rare that they didn’t appear in the gold standard dataset. But for false positives, we could randomly sample images from any consensus classification – so I gathered a bunch of images that had been identified as these rare species and checked them to calculate false positive rates.
Now, if a species has really low rates of false negatives and really low rates of false positives, then it’s one people are really good at identifying all the time. Note that in general, species have pretty low rates of both types of error. Furthermore, species with lower rates of false negatives have higher rates of false positives. There aren’t really any species with high rates of both types of error. Take rhinos, for example: folks often identify a rhino when it’s not actually there, but never miss a rhino if it is there.
Also: we see that rare species are just generally harder to identify correctly than common species. The plot below shows the same false negative and false positive error rates plotted against the total number of pictures for every species. Even though there is some noise, those lines reflect significant trends: in general, the more pictures of an animal, the more often folks get it right!
This makes intuitive sense. It’s really hard to get a good “search image” for something you never see. But also folks are especially excited to see something rare. You can see this if you search the talk pages for “rhino” or “zorilla.” Both of these have high false positive rates, meaning people say it’s a rhino or zorilla when it’s really not. Thus, most of the images that show up tagged as these really rare creatures aren’t.
But that’s okay for the science. Because recall that we can assess how confident we are in an answer based on the evenness score, fraction support, and fraction blanks. Because such critters are so rare, we want to be really sure that those IDs are right — but because those animals are so rare, and because you have high levels of agreement on the vast majority of images, it makes it really easy to review any “uncertain” image that’s been ID’d as a rare species.
Pretty cool, huh?